Monte Carlo simulations for optical radiation transfer in tissue
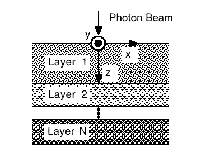 Monte Carlo methods are widely used in simulating the propagation of light in tissue. Monte Carlo methods are widely used in simulating the propagation of light in tissue.
 Consider a laser beam directed onto a tissue sample, as seen in the first graph: as the light propagates through the tissue, some of the photons are reflected back, a small portion of the photons is absorbed and the rest are scattered in random directions. The degree of absorption and scattering of the photons in the tissue depends on the wavelength of the laser light used and the optical and thermal properties of the tissue sample. Each layer has its own set of optical properties such as absorption and scattering coefficients, density, anisotropic factor, and refractive index parameters. Consider a laser beam directed onto a tissue sample, as seen in the first graph: as the light propagates through the tissue, some of the photons are reflected back, a small portion of the photons is absorbed and the rest are scattered in random directions. The degree of absorption and scattering of the photons in the tissue depends on the wavelength of the laser light used and the optical and thermal properties of the tissue sample. Each layer has its own set of optical properties such as absorption and scattering coefficients, density, anisotropic factor, and refractive index parameters.
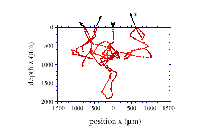
Monte Carlo method follows the random walk of a photon, as depicted in the second graph, until it is absorbed or transmited. By investigating the path that the photons take as they move through the tissue, information about the tissue sample can be obtained. The transmitted photons can be used to locate tumours.
Applications in medicine and current work
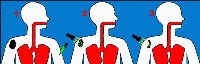 Photodynamic therapy is a light activated chemotherapy. It
is a three step process. Step one is the administering of a photoreactive drug to the cancer patient. After 24-48 hours the dye accumulates in the cancerous cells. Step two involves the activation of this dye with a suitable
laser light. Step three the destruction of the cancer cells. The main side effects of PDT are photosensitivity, which
can last up to four weeks after the time of injection of the fluorescent dye. Exposure to sunlight during this
period results in swelling and redness of the exposed areas. Photodynamic therapy is a light activated chemotherapy. It
is a three step process. Step one is the administering of a photoreactive drug to the cancer patient. After 24-48 hours the dye accumulates in the cancerous cells. Step two involves the activation of this dye with a suitable
laser light. Step three the destruction of the cancer cells. The main side effects of PDT are photosensitivity, which
can last up to four weeks after the time of injection of the fluorescent dye. Exposure to sunlight during this
period results in swelling and redness of the exposed areas.
We are reviewing the current state of Monte Carlo simulations for optical radiation, and updating the MCML simulation code to describe the thermal diffusion in the irradiated tissue. By using a tissue defined array and a grid system, the probability of the photon entering a certain position in the grid can be computed. Finally, we are trying to run this code in parallel so it saves on processing time.
Computed radiography X-ray image processing
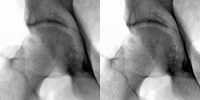 In order to estimate the object image, O(x,y) from the observed image, I(x,y), we use a technique known as regularised deconvolution. It involves iteratively deconvolving the observed image with a measured point spread function, P(x,y). A wavelet denoising algorithm is applied to the residual between iterations. The residual is defined as where On(x,y) is the object estimate at the nth iteration. In order to estimate the object image, O(x,y) from the observed image, I(x,y), we use a technique known as regularised deconvolution. It involves iteratively deconvolving the observed image with a measured point spread function, P(x,y). A wavelet denoising algorithm is applied to the residual between iterations. The residual is defined as where On(x,y) is the object estimate at the nth iteration.
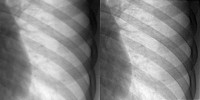 This algorithm provides a better estimate of the object image by reducing the effects due to scatter and noise in the digitised clinical images. This algorithm provides a better estimate of the object image by reducing the effects due to scatter and noise in the digitised clinical images.
We aim to assess image quality based on the judgement of a human observer. Four radiologists assessed the images based on criteria listed in Guidelines on Quality Criteria for Clinical Radiographs. These guidelines have been developed by the Commission for European Communities (CEC, 1996a).
Image criteria:
- Visually sharp reproduction of the vascular pattern in the whole lung
- Visually sharp reproduction of the borders of the heart and aorta
- Visualisation of the retrocardiac lung, spine and mediastinum
|
Method, results and conclusions
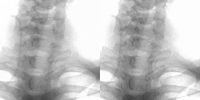 Ten computed radiography PA chest images were randomly chosen from a specific day in a radiology department. Each image was processed using the regularised deconvolution algorithm. The processed and the unprocessed images were placed in a random order and presented to radiologists, who scored five criteria for each image on a rating scale of 1 to 5 (1=poor and 5=excellent). Ten computed radiography PA chest images were randomly chosen from a specific day in a radiology department. Each image was processed using the regularised deconvolution algorithm. The processed and the unprocessed images were placed in a random order and presented to radiologists, who scored five criteria for each image on a rating scale of 1 to 5 (1=poor and 5=excellent).
The results were as follows:
| image criteria |
original image (mean score) |
mean improvement |
standard error |
| vascular lung |
2.6 |
0.5 |
0.3 |
| heart and aorta borders |
3.6 |
0.2 |
0.3 |
| retrocardiac lung, spine & mediastinum |
2.7 |
0.2 |
0.2 |
| contrast |
3.2 |
0.3 |
0.2 |
| noise |
3.4 |
0.6 |
0.2 |
We concluded that the algorithm produces enhanced x-ray images in which we get improved image sharpness and noise quality. These effects are achieved without altering the visibility of anatomical detail. However, the algorithm in computationally intensive and so takes a long time to process each image. We hope to employ this parallel algorithm to configurations with more processors which will reduce processing time.Also, we intend to extend our analysis to include a more appropriate spatially variant PSF. |